Research
Retina
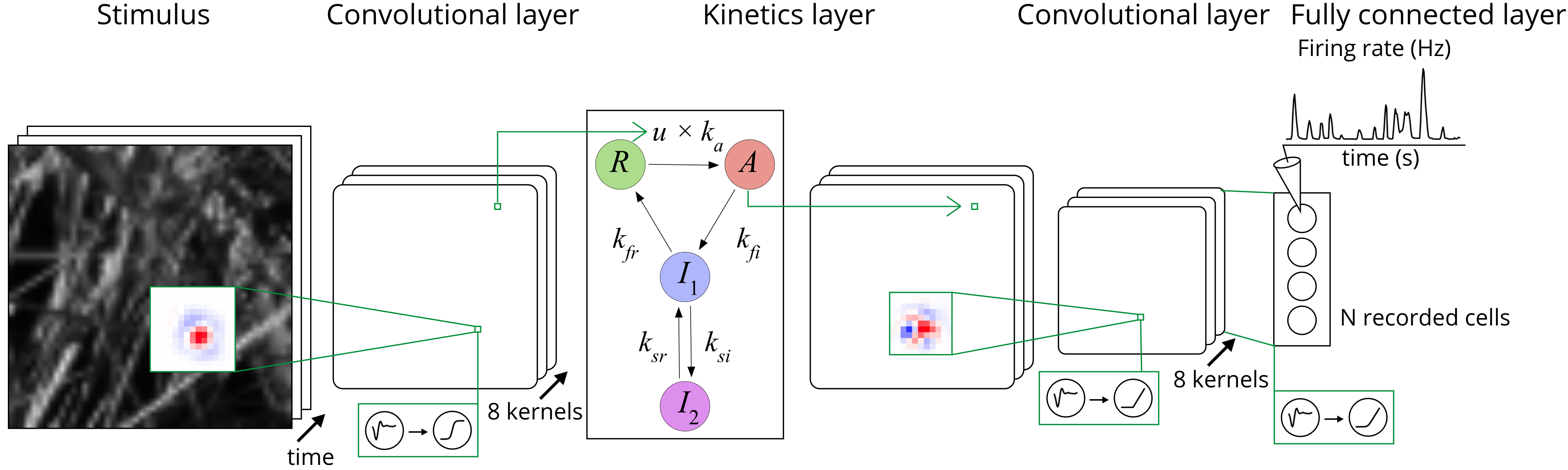
Visual perception is initiated by the molecules, cells, and synapses of the retina, acting together to process and compress visual information into patterns of electrical signals in the optic nerve. Our experimental methods of simultaneous two photon imaging and multielectrode recording allow us unprecedented access to populations of neurons, and recent advantages in machine learning now allow us to accurately model the retinal response to natural scenes. Moreover, these models have internal units that are highly correlated to actual interneurons. Borrowing from methods of interpretable machine learning, we are pioneering new approaches to automatically generate mechanistic circuit hypotheses from neural data for how different cell types combine to create specific neural computations. Testing these hypotheses by recording from and perturbing interneurons is allowing us to give new circuit explanations for ethological computations and their role in natural visual processing.
Cortex
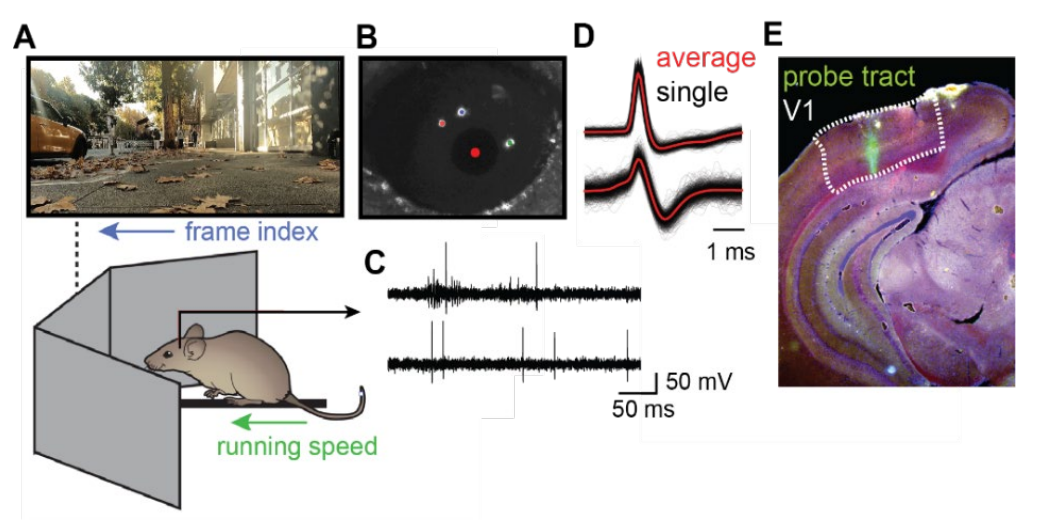
The visual cortex uses a diverse array of cell types to restructure visual information preprocessed by the similarly diverse set of retinal neurons. Although many computations are inherited from the retina such as various types of adaptation, detection and prediction of visual features, others are synthesized de novo. Primary goals are to discover the full set of these computations under natural scenes during visual behavior, and to understand their functional benefits, circuit basis and the underlying theories that drove their evolution. We perform in vivo behavioral experiments in mice in a virtual reality environment and are developing computational models that capture the neural code for natural scenes, relate that code to the responsible neural pathways and cell types. We are analyzing these models to gain insight into how animals sense, learn and act successfully given the statistics of natural scenes.
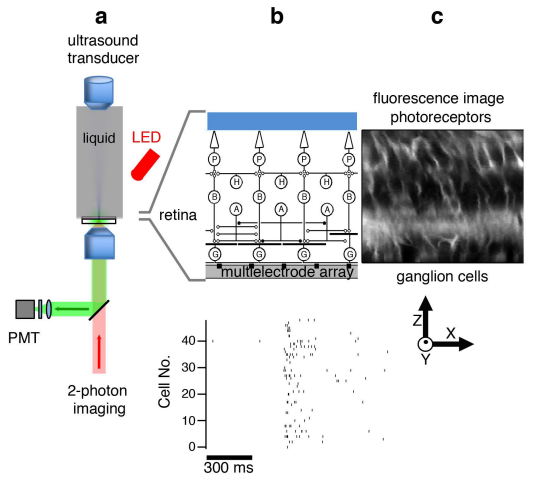
Ultrasound
A critical aspect of understanding complex systems is the ability to perturb components of those systems. Ultrasound has been shown to influence neural activity, and is a potentially useful tool both for basic understanding of the nervous system and a noninvasive treatment for humans in neural disease. In collaboration with electrical and bioengineers, we are developing approaches to stimulate the nervous system with ultrasound. We have shown that ultrasonic neural stimulation is highly precise and reproducible in the in vitro retina but the biophysical mechanisms are unknown. We are using a range of electrophysiological and optical experimental approaches in the retina and hippocampus as well as molecular dynamics simulations to understand the physical and biophysical mechanisms of ultrasonic neurostimulation.